October is American Pharmacists Month
October is American Pharmacists Month and an excellent opportunity for us to celebrate all of our Magellan Rx pharmacy heroes that help our members live healthy, vibrant lives! To kick off American Pharmacists Month, we wanted to shine a spotlight on our two pharmacy residents. We will be taking you behind the scenes to find out what got them interested in pharmacy and what they love most about their residency program.
Magellan Rx: Why did you decide you wanted to become a pharmacist?
Shyra Bias, PharmD: I decided at a young age that I wanted to be the drug expert in my family after witnessing a family member suffer from the complications of extremely uncontrolled latent autoimmune diabetes in adults (LADA). My desire to help my family led to a passion to improve many patients’ lives by ensuring that they can receive the medications they need at a cost they can afford.
Alaka’i Montalbo, PharmD: Growing up, I played a lot of sports and was always in the gym. As an athlete, you learn about sports supplements and how they can help you. In high school, I loved diving into the science of supplements. I later learned in life that the “science” of supplements was not backed by clinical data. After having my dreams crushed, I was working in the hospital and one of the nurses asked me if I had ever considered being a pharmacist? She knew I enjoyed talking about supplements and learning how different medications worked in the body. After researching what pharmacists do, I realized this was exactly what I wanted to do, and I have not looked back since.
Magellan Rx: What advice would you give to someone who wants to become a pharmacist?
Shyra Bias, PharmD: Keep your eye on the prize! The road to PharmD is not an easy one, but if you are truly passionate about the profession, you will get through it. Also, pharmacy is a very small world, so be intentional about making connections because you never know who may be able to change the trajectory of your career.
Alaka’i Montalbo, PharmD: Find joy in your journey. While pharmacy school is a long journey, it will fly by in the grand scheme of life. Don’t give up and never stop believing that you can do it. Don’t fall into the trap of saying to yourself I’m not ready yet. Right now, start making connections with pharmacists and current pharmacy students.
Magellan Rx: What different career paths can you do as a PharmD?
Shyra Bias, PharmD: There are many options for Pharmacists; many do not come to mind immediately when someone thinks of the traditional roles of a pharmacist. Some of the first paths that come to my mind include industry pharmacy, nuclear pharmacy, managed care pharmacy, and clinical pharmacy (hospital setting) to name a few.
Alaka’i Montalbo, PharmD: During pharmacy school, I had the opportunity to go to local high schools and talk about the profession of pharmacy. There are so many different careers Pharmacists can choose and every route is unique. There are traditional roles like retail, hospital pharmacy, and then some non-traditional roles are academia, nuclear (chemotherapy) pharmacy, FDA pharmacists, and Indian health services.
Magellan Rx: What made you choose managed care pharmacy?
Shyra Bias, PharmD: I chose managed care because I really enjoy the population health aspect of the industry. It gives me a true feeling of gratitude to change millions of lives with “the click of a button.”
Alaka’i Montalbo, PharmD: Managed care is the perfect balance between using clinical data and real-world efficacy to help patients manage their disease states. One of my favorite quotes about pharmacy is from Everett Koop, “Drugs don’t work in patients who don’t take them.” I would add to this quote, also with patients who can’t afford them. Managed care is that perfect bridge between both worlds of pharmacy, and I feel like I am making a huge difference every day, not just for one patient but for entire populations.
Magellan Rx: How do you like your residency at Magellan Rx?
Shyra Bias, PharmD: I am grateful beyond words to have been matched with Magellan Rx. The company culture makes me feel extremely welcomed, and the variety of experiences that we are exposed to through our rotations will allow us to succeed no matter where I end up in the managed care industry.
Alaka’i Montalbo, PharmD: Being Native Hawaiian, Ohana (family) means everything to me, and family means no one gets left behind or forgotten. Magellan Rx is one big Ohana and every day, I get to stand on the shoulders of giants. I feel so blessed to be a part of this company and everyone in this residency program is always willing to stop what they are doing, take time out of their busy schedules to help me grow! I cannot express my gratitude enough and I look forward to paying it forward in my career.
Don’t forget to follow us on LinkedIn and Twitter where we will be featuring different pharmacists and pharmacy techs throughout October. #MRxAmericanPharmacistMonth

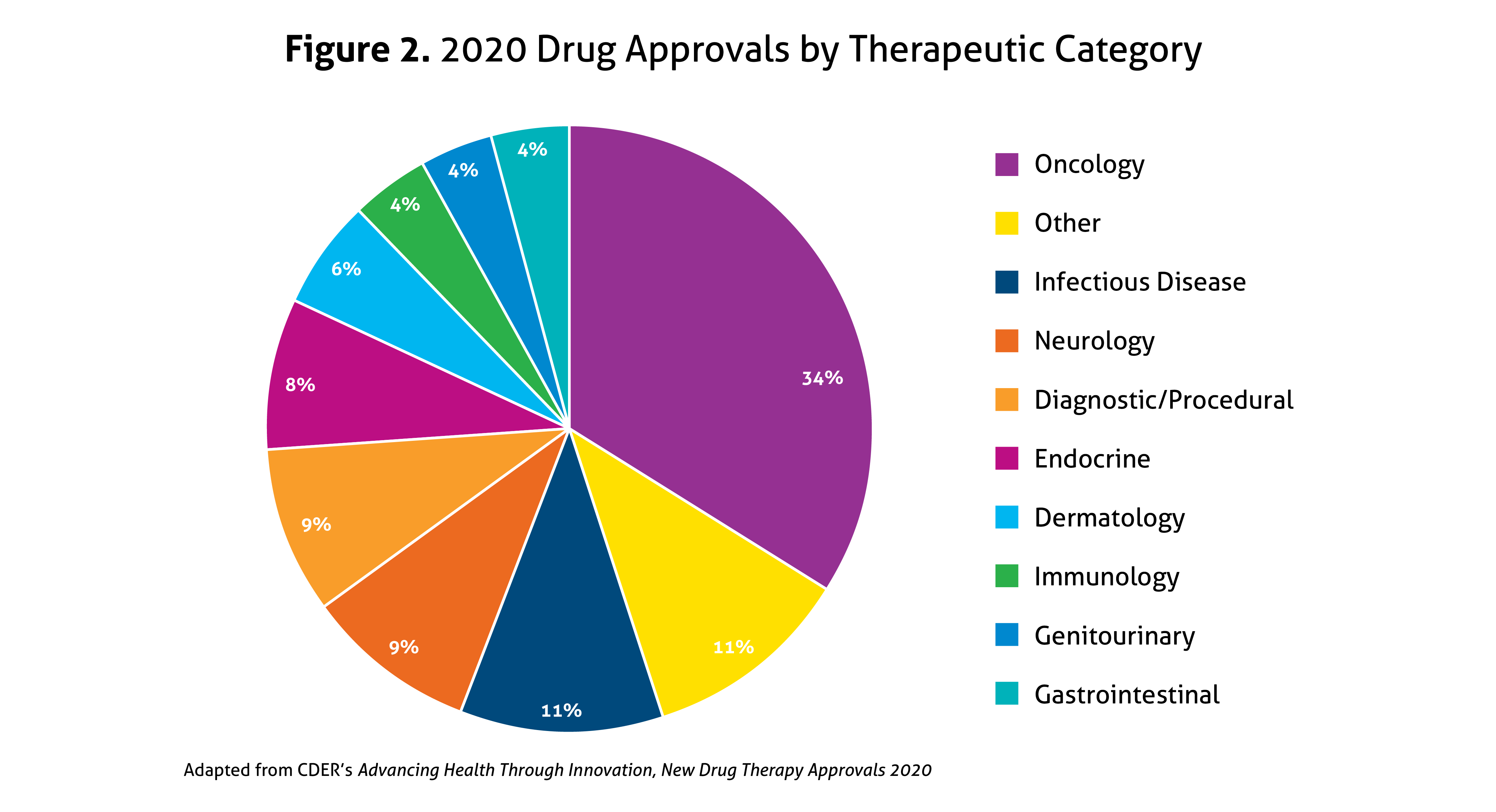
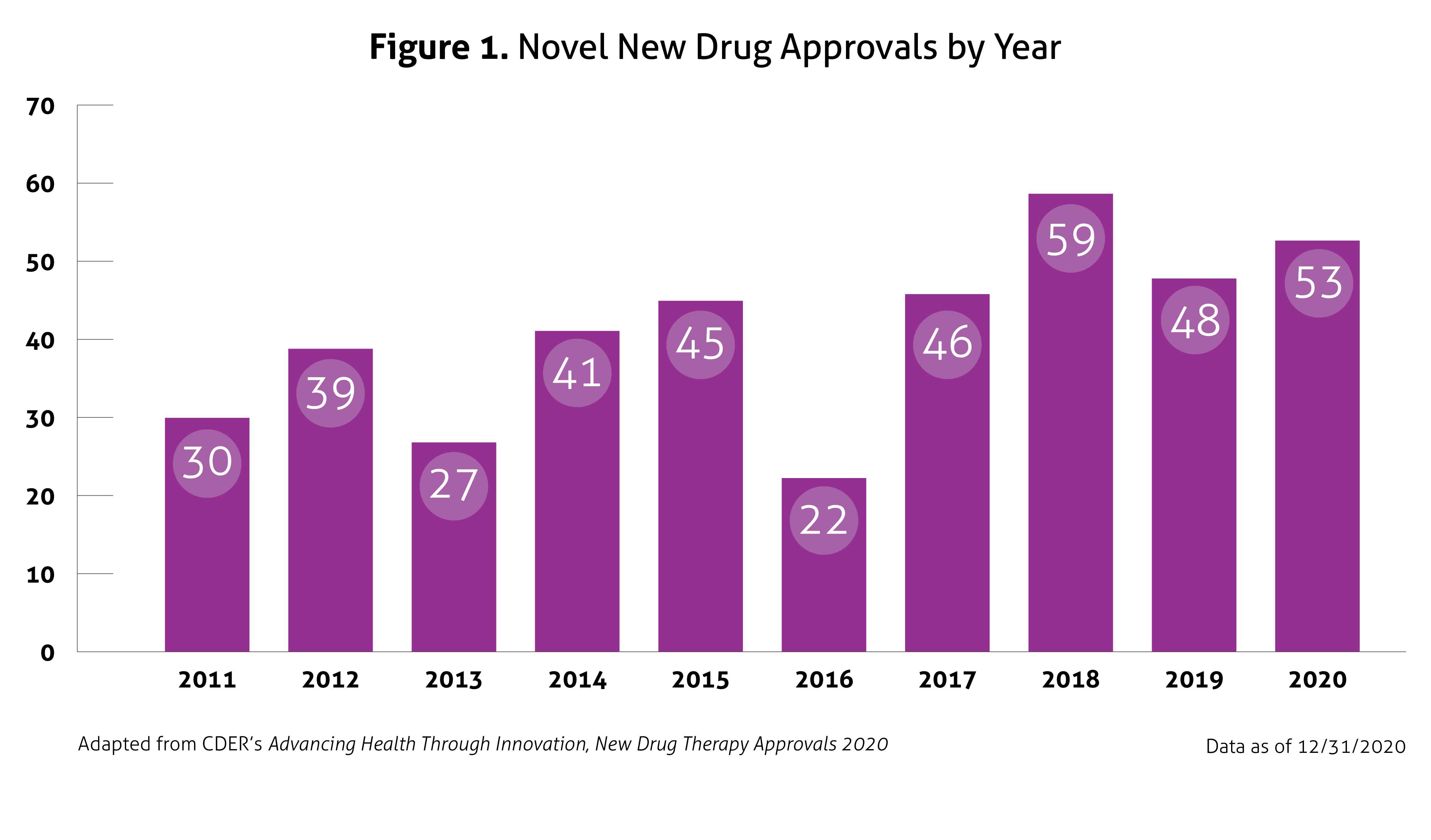 Despite the ongoing pandemic the FDA continued their strategic initiatives to expedite the safe review of treatments in 2020. With the unprecedented challenges incurred in 2020, the FDA acknowledged that maintaining their commitment to bringing forth innovative therapies was difficult. Remarkably, the numbers reported by the FDA do not include the several emergency use authorizations (EUAs) issued by the FDA for COVID-19.
Despite the ongoing pandemic the FDA continued their strategic initiatives to expedite the safe review of treatments in 2020. With the unprecedented challenges incurred in 2020, the FDA acknowledged that maintaining their commitment to bringing forth innovative therapies was difficult. Remarkably, the numbers reported by the FDA do not include the several emergency use authorizations (EUAs) issued by the FDA for COVID-19.