Have you heard that the world of cervical cancer therapy is evolving? There are two players who recently stepped onto the field of oncology for the treatment of recurrent, metastatic, or persistent disease. In 2021, FDA approval moved Keytruda® (pembrolizumab) from second-line to first-line therapy in PD-1 positive patients. Tivdak™ (tisotumab vedotin-tftv) also obtained FDA approval as second-line and subsequent therapy in the same space.
Why is the approval of two therapies in the same year significant? Since the 2014 approval of bevacizumab, no new first-line therapy for cervical cancer has hit the oncology market, and prior to bevacizumab, the last new approval in the category was the combination therapy with topotecan and cisplatin in 2006. So Keytruda and Tivdak are possible game-changers for patients!
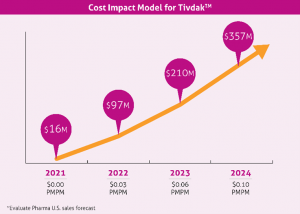
From 1975-2010, new diagnoses of cervical cancer have decreased by more than 50% due to screening. With the introduction of the HPV vaccine in 2006, there was hope that HPV infection prevention would lead to decreased cervical cancer cases. In 2020, a Swedish study published in the New England Journal of Medicine found that the quadrivalent HPV vaccination was associated with a significant reduction in the risk of invasive cervical cancer. In 2018, there were 293,394 women living with cervical cancer in the U.S. As cervical cancer cases decline, according to SEER data, there were an estimated 14,480 new cases and 4,290 deaths reported in 2021. The total annual medical cost of cervical cancer care is estimated to be $1.6 billion. Due to its move from second to first-line, Keytruda will likely demonstrate a net neutral effect on the budget. A forecasted cost impact model for Tivdak is demonstrated below: